Erin Jarvis
Erin joined the lab Fall 2011 through Integrative Biology and graduated Summer 2018.
E-mail: ealberstat (at) mbl.edu
Curriculum Vitae: (CV pdf)
The Evolution of nature’s incredible array of specialized forms, movements, and behaviors—and how the development of each is intertwined with the next—are fascinating yet unresolved topics that I wish to explore.
Research Goals
Homeotic (Hox) genes are important regulators of regional identity along the anterior-posterior (head-to-tail) axis during animal development. Changes to the expression patterns of Hox genes over evolutionary time are thought to be a major driver in the evolution of appendage diversity in arthropods, as suggested by comparative studies such as Averof and Patel, 1997, which correlates phylogenetic shifts in the anterior expression boundary of the Hox gene Ultrabithorax (Ubx) to the occurrence of specialized feeding appendages called maxillipeds. Experimental knockdown and misexpression of Ubx in the amphipod crustacean Parhyale hawaiensis support this claim by recapitulating the phylogenetically observed transformations (Liubicich et al., 2009, Pavlopoulos, et al., 2009). A key missing link (and criticism) to the proposed role of Hox genes in evolutionary transitions, however, is the lack of evidence for the functional integration of these transformed appendages. Specifically, do Hox genes mediate the segment-specific neural and muscular integration required of a functional morphological change?
To answer this questions and others, I perform CRISPR/Cas9 mutagenesis in the emerging model Parhyale hawaiensis, an amphipod crustacean that boasts a great diversity of specialized appendages along its bauplan, as well as good phylogenetic positioning among other crustaceans that constitute an array of appendage diversities for which to speculate evolutionary transitions. My main research goals are to demonstrate identity-specific neuromuscular integration by Hox genes, and to further explore the combinatorial interactions among Hox genes in establishing segment and appendage identities in Parhyale, which deviates from the canonical model of posterior prevalence established by Drosophila studies.
Genetic Interactions among Hox genes in establishing appendage identities
Classical studies in Drosophila demonstrate that Hox gene KO (e.g., Ubx, abdominal A (abd-A), or Abdominal B (Abd-B)) results in all of the affected limbs transforming to the next-most anterior identity. This suggests a model of posterior prevalence, in which the more posteriorly expressed Hox gene exhibits functional dominance, thereby dictating appendage identity in overlapping domains. In Parhyale, however, we demonstrate “skips” in the identities of limbs transformed by abd-A and Abd-B KO indicative of a combinatorial model or “Hox code” (Martin et al., 2016). My project digs deeper into the genetic interactions among Hox genes in establishing limb identity, revealing the shifts in Hox expression domains induced by the knockout of neighboring Hox genes. I demonstrate the homeotic transformations that result from knocking out multiple Hox genes in a single animal (e.g., abd-A + Abd-B, Ubx + Abd-B), and show the actual, induced Hox patterns that underly these transformed identities.
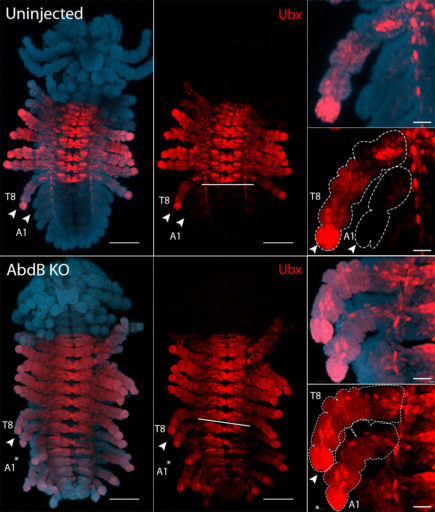
Abd-B represses the expression of Ubx in Parhyale
Abd-B KO (expressed throughout the abdomen in WT, not shown) results in the expression of Ubx (restricted to the thorax in WT) to extend throughout the abdomen.
Segment-specific tissue integration by Hox genes
My main research goal is to demonstrate the functional integration of regional tissue identities mediated by Hox genes by characterizing the segment-specific molecular expression of neural and muscle patterning during embryonic development and the resulting neural wiring and musculature that underlie the morphological transformation of appendage identity induced by Abd-B knockout.
Parhyale undergo holoblastic cleavage, and lineage-tracing studies reveal that each blastomere is tissue-restricted at the 8-cell stage, allowing me to specifically target either a mesoderm or ectoderm lineage for gene knockout by CRISPR/Cas9 microinjection. This enables me to address the interplay between germ layers, and whether genetic changes to either the ectoderm or mesoderm can drive changes in the other, promoting a system of internal stabilization that maintains coordination between structures. Studies in Drosophila suggest that changes to the ectoderm can coordinate some, but not all, changes to the mesoderm (Dutta, et al 2010), and the direct development of Parhyale may reveal even greater coordination. Demonstration of an interconnected development program that provides functional robustness will further supports the idea that small changes in regulatory genes may affect complete changes in morphology.
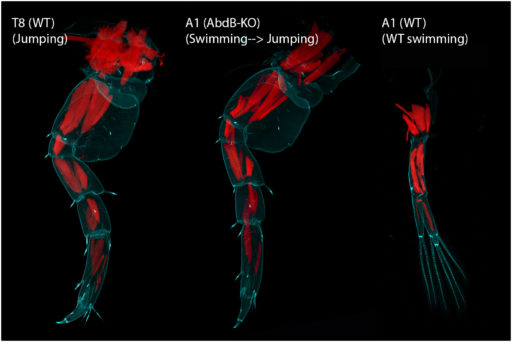
Abd-B KO gives a near complete transformation of the underlying musculature
(Cyan = autofluorescence, Red = Phalloidin)
Publications
Martin A, Serano JM, Jarvis E, Bruce HS, Wang J, Ray S, Barker CA, O’Connell LC, Patel NH (2016) CRISPR/Cas9 Mutagenesis Reveals Versatile Roles of Hox Genes in Crustacean Limb Specification and Evolution. Current biology 26:14–26.
Serano JM, Martin A, Liubicich DM, Jarvis E, Bruce HS, La K, Browne WE, Grimwood J, Patel NH (2016) Comprehensive analysis of Hox gene expression in the amphipod crustacean Parhyale hawaiensis. Developmental biology 409:297–309.
Kao D, et al. (In press) The genome of the crustacean Parhyale hawaiensis: a model for animal development, regeneration, immunity and lignocellulose digestion. E-life
Dalton D, Smit-Robinson H, Vermaak E, Jarvis E, Kotze A (submitted) Is there genetic connectivity among the Critically Endangered Whited-winged Flufftail (Sarothrura ayresi) populations from South Africa and Ethiopia? African Journal of Ecology.
An DD, Kullgren B, Jarvis E, Abergel RJ (2016) From early prophylaxis to delayed treatment: Establishing the plutonium decorporation activity window of hydroxypyridinonate chelating agents. Chemico-biological interactions (Article In Press).
Sturzbecher-Hoehne M, Kullgren B, Jarvis E, An DD, Abergel RJ (2014) Highly luminescent and stable hydroxypyridinonate complexes: a step towards new curium decontamination strategies. Chemistry 20:9962–9968.
Nijagal A, Derderian C, Le T, Jarvis E, Nguyen L, Tang Q, Mackenzie TC (2013) Direct and indirect antigen presentation lead to deletion of donor-specific T cells after in utero hematopoietic cell transplantation in mice. Blood. 121:4595-602.
Kullgren B, Jarvis E, An DD, Abergel RJ (2013) Actinide chelation: biodistribution and in vivo complex stability of the targeted metal ions. Toxicology mechanisms and methods 23:18–26.
Bunin DI, Chang PY, Doppalapudi RS, Riccio ES, An D, Jarvis E, Kullgren B, Abergel RJ (2013) Dose-dependent efficacy and safety toxicology of hydroxypyridinonate actinide decorporation agents in rodents: towards a safe and effective human dosing regimen. Radiation research 179:171–182.
Jarvis E, Bruce HS, Patel NH (2012) Evolving specialization of the arthropod nervous system. Proceedings of the National Academy of Sciences 109:10634–10639.
Jarvis EE, An DD, Kullgren B, Abergel RJ (2012) Significance of Single Variables in Defining Adequate Animal Models to Assess the Efficacy of New Radionuclide Decorporation Agents: Using the Contamination Dose as an Example. Drug Dev Res 73:281–289. (Jarvis 2012)
Osterland CD, Jarvis E, Chadayammuri A, Unnithan R, Weiser MJ, Spencer RL. (2011) Tonic, But Not Phasic Corticosterone, Constrains Stress ActivatedExtracellular-Regulated-Kinase 1/ 2 Immunoreactivity Within the Hypothalamic Paraventricular Nucleus. Journal of Neuroendocrinology 23:1241–1251.
Nijagal A, Wegorzewska M, Jarvis E, Le T, Qizhi T, MacKenzie TC. (2011) Maternal T cells limit engraftment after in utero hematopoietic cell transplantation in mice. J Clin Invest 121:582–592.
Pace TWW, Gaylord RI, Jarvis E, Girotti M, Spencer RL (2009) Differential glucocorticoid effects on stress-induced gene expression in the paraventricular nucleus of the hypothalamus and ACTH secretion in the rat. Stress 12:400–411.


